chapter 15: air pollution
Big ideas: air is shared by everyone on the planet, crosses international borders without consent, impacts innocents with little power.
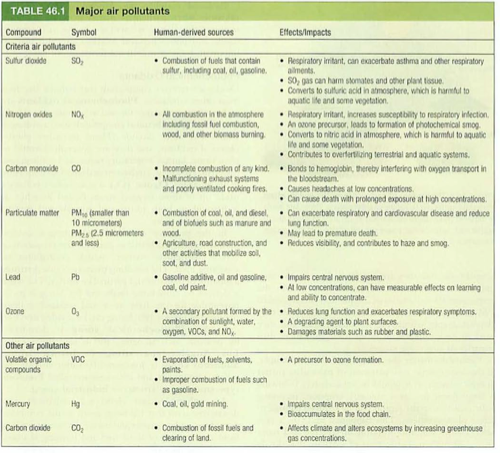
6 major pollutants: “criteria pollutants”
Sulfur dioxide SO2
Nitrogen oxides NOx
Carbon monoxide CO
Particulate matter PM
Ozone O3
Lead Pb
mnemonic: SOX/NOX, CO/O3, PM/Pb
add: CO2 (2007), VOC, Hg
SO2
From Methionine (amino acid in living things) in fossil fuels
Combines with water in the air to make H2SO4 sulfuric acid
NOx
Various forms of oxidized nitrogen, which alone is inert
From high temperature combustion (e.g. auto engines)
Also from decomposition of fertilizers
CO
Emission from car exhaust, or other incomplete combustion:
C + O2 —> CO2
If not enough O2, then CO forms
Toxic, causes damage to hemoglobin, 30 day life cycle for hemoglobin
See also CO2 and climate change, > 400ppm since 2012
PM
PM2.5 is most dangerous, smaller particles lodge deep in the lungs, beyond ability of pulmonary cilia to flush out
PM5, PM10 larger particles
Main cause: power plants, diesel engines
See also vog: PM2.5 particles of ash with SO2 dissolved in water droplets: physical abrasive + corrosive acid = respiratory damage
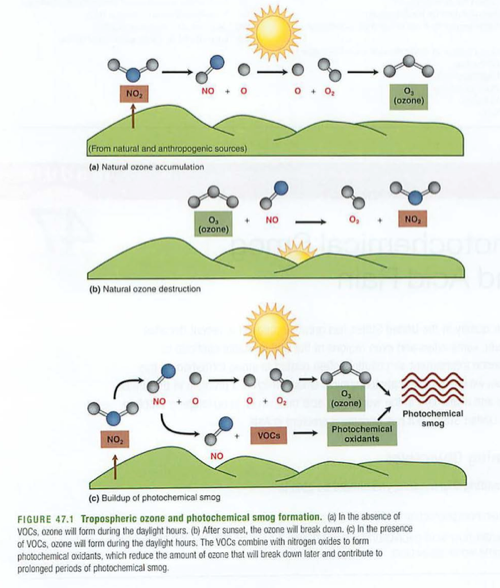
Ozone and other photochemical oxidants (PANs)
Ozone in stratosphere is good, in troposphere bad
Causes lung irritation, toxic to some organisms
Smog and other photochemical reactions:
Pb
Gasoline additive (tetraethyl lead, improves octane rating cheaply), replaced by other worse carcinogenic (cancer causing) chemicals like MTBE:
https://en.wikipedia.org/wiki/Methyl_tert-butyl_ether
Decreases mental capacity (e.g. Roman insanity theory)
Found in coal smoke, along with Mercury and other heavy metals. Sludge from mining is worse.
VOC
Volatile organic compounds, e.g. gasoline vapors (why your gas cap must be on or you get a dashboard warning):
https://en.wikipedia.org/wiki/Volatile_organic_compound
Secondary pollutants: need a chemical reaction (often energy from sunlight) to form (see figure 47.1 above)
See PANs: Peroxyacyl nitrates: formed from VOCs, NOx
https://en.wikipedia.org/wiki/Peroxyacyl_nitrates
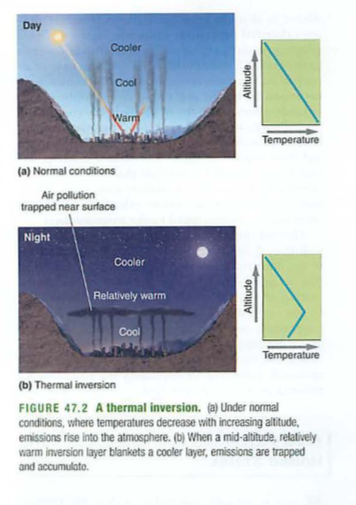
See thermal inversions: london fog, US
Donora, PA 1948 k.20, sick 7000
1952 London 4000-12,000 dead, three nights
Mexico city 1996 300 dead, 400K sick
acid rain: plants, fish, structures
SO2 + O2, water = H2SO4 (sulfuric acid)
NO3 + O2, water = HNO3 (nitric acid)
“Acid snow”

W. VA had rain more acidic than stomach acid
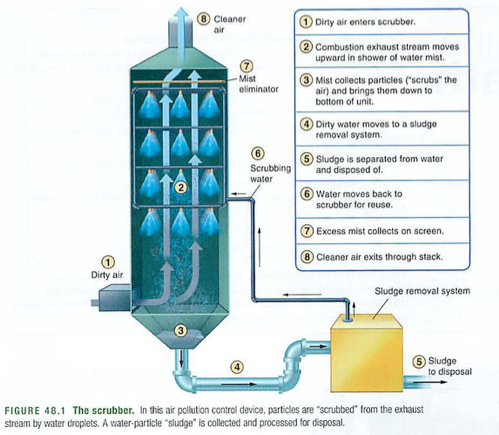
know how a scrubber works (just like the nuclear plant diagram)
IAP-indoor air pollution
Leading cause of death in LDC (women)
manure, open pit fires, CO, PM10
sick building syndrome: formaldehyde, CO2, VOC
the main culprits:
VOC, CO, Radon, Lead, formaldehyde, asbestos, PM10
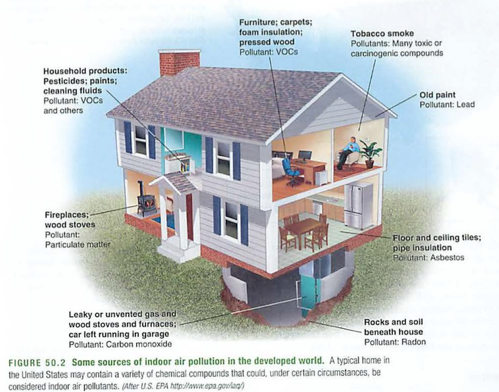
asbestos-asbestosis, mesothelioma
Radon222-lung cancer (smoke demo-lungs)

AIR QUALITY LABS
• Air Quality: Air quality can be assessed using various
methods.
• Particulates: Sticky paper can be used to collect
air particulates from various sources, and then the
paper can be examined under a microscope. It is
not possible to see the smallest particulates, but
they do color the white paper.
• Ozone: In this lab, an ecobadge or a homemade
potassium iodide gel sampler is hung or worn in
order to collect data on tropospheric ozone. The
badge or KI sample changes color in the presence
of ozone and becomes more intensely colored as
the amount of ozone increases.
• Carbon dioxide: In this lab, a commercial sampling
device is used to determine the amount of
carbon dioxide in an air sample. Car exhaust,
burning tobacco, or other pollutants can also be
sampled.
Pollution roundup:
Chapter 8 of Princeton Review for AP:
http://physics.hpa.edu/physics/apenvsci/apes_princeton/ch08-pollution.pdf
http://physics.hpa.edu/physics/apenvsci/apes-5-5/18-pollution.pdf
Comments