
apes unit 2: systems
Chapter test:

Chapter link:
http://physics.hpa.edu/physics/apenvsci/cc15/ch02.pdf
Notes:
Arcata as an ecosystem
closed/open systems
throughput: total in and out
pos feedback: disturbance gets greater
neg feedback: stability restored (equilibrium, homeostasis: same state)
Ship example: cm over cb, = not stable, cb over cm = stable
n.b. fig 2.3: keep in mind eutrophication
resilience: ability to withstand small perturbations
emergent systems: greater than the sum of the separate parts
matter: space and mass
cons of matter
elements: atom (no cuts)
atomic number
atomic mass
isotopes
n,p,e
mainly empty space (stadium example)
elements
compounds
ionic: electrons traded (e.g. salt, high mp, conducts electricity)
covalent: shared electrons (e.g. plastic, butter, low mp, insulation)
bonds: stored energy
breaking bonds takes energy, forming releases energy:
burning wood: many smaller bonds formed (CO2), releases energy
OILRIG: oxidation is loss of electrons, reduction is gain
Periodic table: stability, ions: cations and anions
Acids and bases: acids donate H+ (proton donor), bases accept H+ (proton acceptor)
Many definitions of acids and bases…
Common acids: HCl, H2SO4, HNO3, H3PO4
Common bases: NaOH, KOH, Ca(OH)2
pH scale: 7 neutral
pH = log10 [H+] see examples
n.b. water planet section:
Polar covalent: universal solvent
Liquid at 0-100°C
cohesion and adhesion, t.f. capillary action
expands when it freezes, t.f. solid is less dense than liquid, floats: preserves life over winters (4°C rule)
540 cal/g Qv, useful for heat transfer (sweating, evaporation)
c = 1.00 cal/g°C, very high specific heat, ability to stabilize heat energy, act as storage (thermal inertia)
Organic compounds: contain carbon
Bio-organics: lipids, carbohydrates, proteins, nucleic acids
lipids: CH chains, not water soluble, see also glycol backbones
all single bonds (fully saturated with H), easy to fit together, hard to break (melt), so these are solids (fats)
some double bonds (unsaturated), easier to melt, these are oils
carbohydrates: CH chains with OH replacements, water soluble
simple chains are called sugars, complex chains are starches, see also glycol backbones, triglycerides
proteins: CHO + N
all hormones, most structures, elements of nucleotides
Nucleotides: PO4 group + protein (CHON) + sugar (ribose or deoxyribose)
only four proteins used in DNA: A = adenine, C = cytosine, G = guanine, T = tyrosine
C+G T+A elegant pairs
Cellular terms:
Phospholipid bilayer: lipids, proteins, channels, gateways, pumps
mitochondria: O2 metabolism
Nutrients: NPK
NItrates NO3
Phosphates PO4
K (potassium, potash = K2O)
Energy:
kinetic-motion (kinesias) 1/2mv2
potential-usually gravitational potential energy (ugh)
chemical energy-usually bonds, ability to be metabolized (oxidized)
Heat: thermal energy
calories, 1 cal = 4.18 joules
Heat always moves from hot to cold, harnessing this motion is called a heat engine (auto, hurricane)
To move heat uphill, this requires energy (air conditioner)
High quality energy: concentrated (e.g. oil)
Low quality energy: diffuse (e.g. wind, ocean thermal)
Thermodynamics: physics version
1. you can't win
2. you can't break even
3. you can't get out of the game
Bio versions of these:
1. all energy is conserved, may be transformed, but never lost or created
2. each transaction produces an energy tax: no reaction is 100% efficient, and all reactions tend towards disorder (entropy) and the low quality heat death of the universe
Most heat engines (autos, humans) are about 30-40% efficient. Most is the diesel engine at about 60%
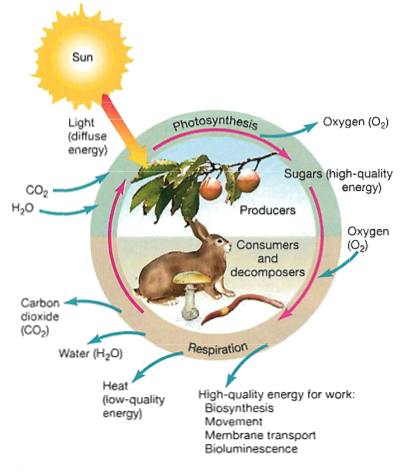
Solar energy direct to plants: photosynthesis (primary producer)
exception: thermal energy is chemosynthesis (sulfur ports, submarine nuclear reactors)
Above 40°C most proteins are denatured (break apart), so enzymes are needed (catalysts)
Enzymes reduce activation energy (threshold) of reactions
Every 10°C increase or decrease in temp doubles/halves the reaction rate (e.g. fever)
Photosynthesis
about 1500 W/m2 arrives on planet, some reflected by clouds, dust, atmosphere
remaining energy is absorbed by chlorophyll and other -phylls. chlorophyll is mainly red and blue (looks green as a result)
Fall = chlorophyll activity decreases, remaining -phylls are still active (carotenes, xanthophylls, etc.)
http://en.wikipedia.org/wiki/Photosynthesis
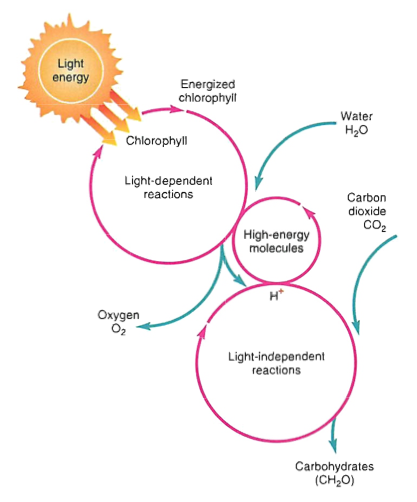
n.b. light dependent reactions (chlorophylls in the chloroplasts)
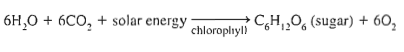
enzymes split water into H and O, releasing the oxygen, create ATP and NADPH (high energy molecules)
another set of enzymes combine carbon from CO2 to form sugars.
Plant photosynthesis
Reverse reaction happens at night, so plants don't die when the sun sets (cellular respiration):
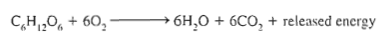
Recall that Maui onions are sweet: lots of sunlight in the daytime, onions store sugar. Nights are cool, slowing metabolism, so the sugar builds up (they store more than they use at night). Result: sweet onions
Species: Same kind, genetically similar so they can breed
Population: same species same place, same time
Many species populations make up a community
Communities make up an ecosystem
productivity = amount of biomass produced
low NPP (net primary productivity) in an ecosystem could be due to many detritovores decomposing the biomass
Food webs: complex inter-relationships between organisms that are sustainable, stable and cooperative
Trophic (food) levels:
Primary producers: plants
Primary consumers: eat plants
Secondary consumers: eat the plant eaters (carnivores)
Tertiary consumers: apex predators
See also scavengers, detritovores
Pyramids: 10% of energy passes to next higher level (meat eaters only get 1/100 of original energy, or less, depending on the primary sources)
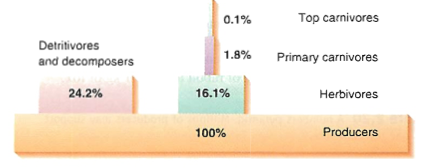
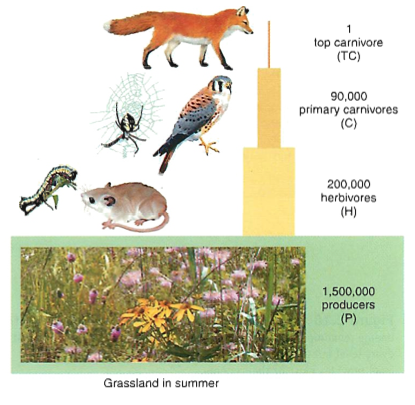
Cycles
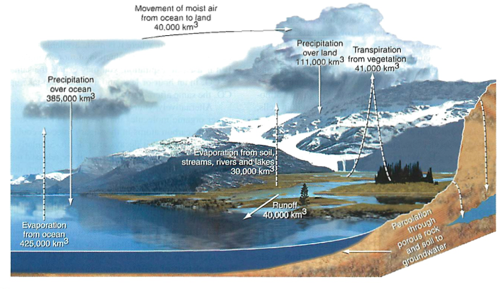
Water cycle: (yes, there is more)
Most water is stored in the oceans (duh)
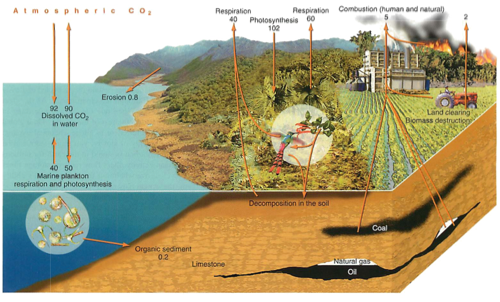
Carbon cycle:
sources and sinks
most is stored as CO3 in rocks, or in the oceans
remember: solids are much denser than liquids
Nitrogen Cycle: Most stored in the air (nitrogen gas)
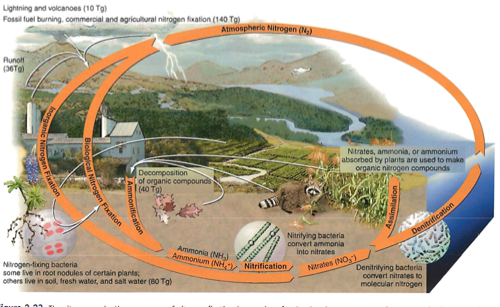
Phosphorus Cycle:
stored in rocks

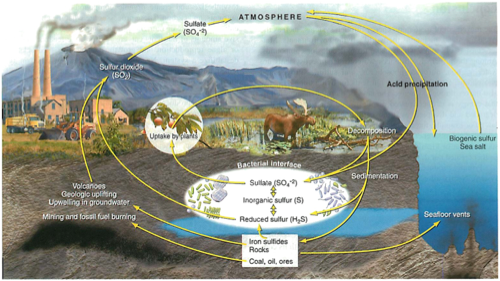
Sulfur cycle:
Most as rocks (pyrite and gypsum=drywall)

Comments